
Custom 3D printed replica hearts look and pump just like the real thing
MIT engineers hope to help doctors adapt treatment to a patient’s special heart shape and function, with a special robotic heart. The team has developed a procedure to 3D print a soft and flexible replica of a patient’s liver. Image: Melanie Gonick, MIT
By Jennifer Chu | MIT News Agency
No two hearts beat the same. The size and shape of the heart can vary from person to person. This difference can be seen most clearly in people living with heart disease, because the heart and their major blood vessels work harder to cope with impaired function.
MIT engineers hope to help doctors adapt treatment to a patient’s special heart shape and function, with a special robotic heart. The team has developed a procedure to 3D print a soft and flexible replica of a patient’s liver. They could then control the replica’s actions to mimic that patient’s blood-pumping ability.
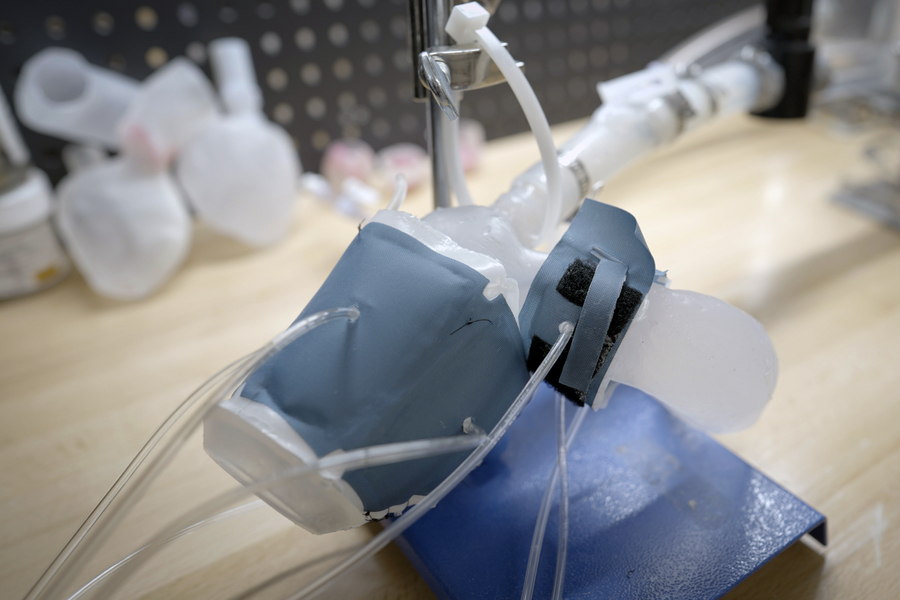
The procedure involves first converting medical images of a patient’s heart into a three-dimensional computer model, which the researchers can then 3D print using a polymer-based ink. The result is a soft, flexible shell that is shaped exactly like the patient’s own heart. The team can also use this approach to print a patient’s aorta — the main artery that carries blood out of the heart to the rest of the body.
To mimic the pumping action of the heart, the team has created a sleeve similar to a blood pressure cuff that wraps around the printed heart and aorta. The bottom of each sleeve resembles bubble wrap in the right pattern. When the sleeve was connected to a pneumatic system, researchers could tune the escaping air to rhythmically inflate the sleeve bubble and contract the heart, mimicking its pumping action.
Researchers can also develop separate sleeves that surround the printed aorta to constrict the vessels. This narrowing, they say, can be tuned to mimic aortic stenosis — a condition in which the aortic valve narrows, causing the heart to work harder to force blood around the body.
Doctors usually treat aortic stenosis by implanting a synthetic valve designed to widen the aortic’s natural valve. In the future, the team says doctors could potentially use their new procedure to first print a patient’s heart and aorta, then implant various valves into the molded model to see which design functions best and is a good fit for that patient. Heart replicas can also be used by research laboratories and the medical device industry as realistic platforms for testing therapies for various types of heart disease.
“All hearts are different,” said Luca Rosalia, a graduate student in the MIT-Harvard Program in Health Sciences and Technology. “There is a huge variation, especially when the patient is sick. The advantage of our system is that we can not only recreate the shape of a patient’s heart, but also its function in physiology and disease.”
Rosalia and his colleagues report their results in a study that appears in Science Robotics. MIT co-authors include Caglar Ozturk, Debkalpa Goswami, Jean Bonnemain, Sophie Wang, and EllenRochealong with Benjamin Bonner of Massachusetts General Hospital, James Weaver of Harvard University, and Christopher Nguyen, Rishi Puri, and Samir Kapadia at the Cleveland Clinic in Ohio.
Print and pump
In January 2020, team members led by mechanical engineering professor Ellen Roche developed a “heart of biorobotic hybrid” — a common replica of the heart, made of synthetic muscle containing tiny inflatable cylinders, which they can control to mimic the contractions of a real beating heart.
Shortly after that effort, the Covid-19 pandemic forced Roche’s lab, along with most others on campus, to temporarily close. Undeterred, Rosalia continues to tweak the home’s heart-pumping designs.
“I rebuilt the entire system in my dorm room that March,” recalls Rosalia.
Several months later, the lab reopened, and the team picked up where it left off, working to improve heart-pumping sleeve control, which they tested in animals and computing model. They then expanded their approach to develop replicas of arms and hearts that were specific to each patient. For this, they turned to 3D printing.
“There is a lot of interest in the medical field in using 3D printing technology to accurately recreate patient anatomy for use in preprocedure planning and training,” notes Wang, who is a resident of vascular surgery at Beth Israel Deaconess Medical Center in Boston.
Inclusive design
In the new study, the team utilized 3D printing to produce custom replicas of real patient hearts. They use a polymer-based ink that, once printed and cured, can be squeezed and stretched, similar to a real heartbeat.
As source material, the researchers used medical scans of 15 patients diagnosed with aortic stenosis. The team converted each patient’s image into a three-dimensional computer model of the patient’s left ventricle (the main pumping chamber of the heart) and the aorta. They fed these models into a 3D printer to produce soft, anatomically accurate shells of the ventricles and blood vessels.
The actions of the soft robotic model can be controlled to mimic the patient’s pumping ability. Image: Melanie Gonick, MIT
The team also made sleeves to wrap printed forms. They adjust each sleeve pocket in such a way that, when wrapped around the individual shape and connected to a small air pumping system, the sleeve can be adjusted individually to realistically shrink and constrict the printed model.
The researchers demonstrated that for each heart model, they could accurately recreate the same heart pumping pressure and flow that was previously measured in each patient.
“Being able to adjust the patient’s flow and pressure is very encouraging,” says Roche. “We not only printed the heart’s anatomy, but also replicated its mechanics and physiology. That’s the part that gets us excited.”
Going a step further, the team aims to replicate some of the interventions some patients have had, to see if the printed heart and blood vessels respond in the same way. Some patients have received valve implants designed to widen the aorta. Roche and his colleagues implanted similar valves in aortic casts that were modeled after each patient. When they enabled the printed heart to pump, they observed that the implanted valve produced just as good a flow as in the actual patient after their surgical implant.
Finally, the team used the imprinted heart that was moved to compare implants of different sizes, to see which would produce the best fit and flow – something they envision doctors could potentially do for their patients in the future.
“Patients will have their imaging completed, which they do anyway, and we will use it to build this system, ideally within a day,” said co-author Nguyen. “Once it’s up and running, doctors can test different types and sizes of valves and see which works best, then use that for the implant.”
Ultimately, Roche says patient-specific replicas can help develop and identify the ideal treatment for individuals with unique and challenging cardiac geometries.
“Designing inclusively for a large number of anatomies, and testing interventions across these ranges, can increase the addressable target population for minimally invasive procedures,” said Roche.
This research was supported in part by the National Science Foundation, the National Institutes of Health, and the National Heart Lung Blood Institute.
tags: c-Health-Medicine


MIT News






