Researchers designed a new system to convert seawater into hydrogen fuel
[ad_1]
(Nanowerk News) The mixture of seawater consisting of hydrogen, oxygen, sodium and other elements makes it vital for life on Earth. But the same complex chemistry has made it difficult to extract hydrogen gas for clean energy use.
Now, researchers at the Department of Energy’s SLAC National Accelerator Laboratory and Stanford University with collaborators at the University of Oregon and Manchester Metropolitan University have found a way to remove hydrogen from the oceans by flowing seawater through a double-membrane system and electricity. Their innovative design proved successful in producing hydrogen gas without producing large amounts of harmful by-products. The results of their study, published in Joules (“Hydrogen production with a seawater-resistant bipolar membrane electrolyzer”), could help advance efforts to produce low-carbon fuels.
“Many water-to-hydrogen systems today try to use single-layer or single-layer membranes. Our study brings the two layers together,” said Adam Nielander, an associate staff scientist with the SUNCAT Center for Interface Science and Catalysis, a joint SLAC-Stanford institute. “This membrane architecture allows us to control the way the ions in seawater move in our experiments.”
Hydrogen gas is a low-carbon fuel currently used in many ways, such as to power fuel cell electric vehicles and as a long-term energy storage option – one suitable for storing energy for weeks, months or longer – for the power grid.
Many attempts to make hydrogen gas start with fresh water or desalination, but these methods can be expensive and energy intensive. Treated water is easier to work with because it has fewer ingredients – chemical elements or molecules – floating around. However, purifying water is expensive, requires energy and adds complexity to the device, the researchers said. Another option, natural fresh water, also contains a number of impurities that are problematic for modern technology, in addition to being a more limited resource on the planet, they said.
To work with seawater, the team applied a bipolar, or two-layer, membrane system and tested it using electrolysis, a method that uses electricity to move ions, or charged elements, to carry out the desired reactions. They began their design by controlling the most dangerous element to the seawater system – chloride – said Joseph Perryman, a SLAC and Stanford postdoctoral researcher.
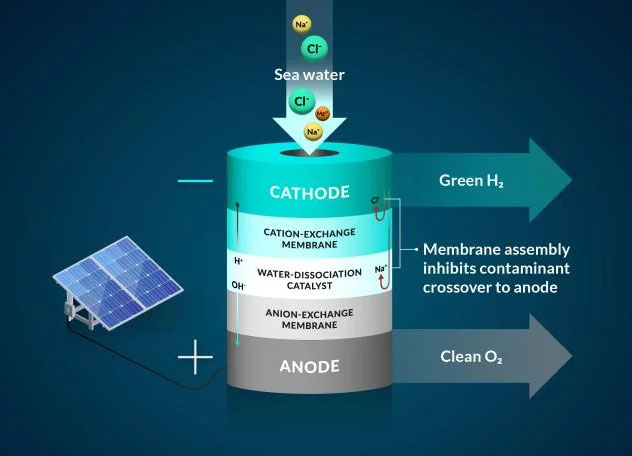
“There are many reactive species in seawater that can interfere with the water-to-hydrogen reaction, and sodium chloride, which makes seawater salty, is one of the main culprits,” Perryman said. “In particular, chlorides reaching the anode and being oxidized will reduce the life of the electrolysis system and can actually be unsafe due to the toxic nature of the oxidation products which include molecular chlorine and bleach.”
The bipolar membrane in the experiment allows access to the conditions necessary to create hydrogen gas and reduce chloride from reaching the reaction center.
“We’re basically doubling down on the way to stop this chloride reaction,” Perryman said.
Home for hydrogen
An ideal membrane system performs three main functions: it separates hydrogen and oxygen gases from seawater; helps remove only useful hydrogen and hydroxide ions while limiting other seawater ions; and help prevent unwanted reactions. Capturing these three functions together was difficult, and the team’s research was targeted at exploring systems that could efficiently combine these three needs.
Specifically in their experiment, protons, which are positive hydrogen ions, pass through one of the layers of the membrane to a place where they can be collected and converted into hydrogen gas by interacting with a negatively charged electrode, or cathode. The second membrane in the system only allows negative ions, such as chloride, to pass through.
For added support, one layer of the membrane contains negatively charged groups attached to the membrane, which make it difficult for other negatively charged ions, such as chloride, to go where they shouldn’t, said Daniela Marin, Stanford graduate chemical engineering student and co-author. The negatively charged membrane proved highly efficient at blocking nearly all chloride ions in the team’s experiments, and their system operated without producing toxic byproducts such as bleach and chlorine.
Along with designing a seawater-to-hydrogen membrane system, the research also provides a better general understanding of how seawater ions move through the membrane, the researchers said. This knowledge can help scientists design more robust membranes for other applications as well, such as generating oxygen gas.
“There is also interest in using electrolysis to produce oxygen,” said Marin. “Understanding ion flow and conversion in our bipolar membrane system is also very important for this endeavor. Along with hydrogen production in our experiments, we also show how to use a bipolar membrane to generate oxygen gas.”
Next, the team plans to improve their electrodes and membrane by building them with more abundant, more easily mineable materials. These design improvements could make electrolysis systems easier to adapt to the size needed to produce hydrogen for energy-intensive activities, such as the transportation sector, the team said.
The researchers also hope to bring their electrolysis cell to SLAC’s Stanford Synchrotron Radiation Lightsource (SSRL), where they can study the atomic structure of catalysts and membranes using the intense X-ray facility.
“The future is bright for green hydrogen technology,” said Thomas Jaramillo, professor at SLAC and Stanford and director of SUNCAT. “The fundamental insights we gained are key to informing future innovations for the increased performance, durability, and scalability of this technology.”
[ad_2]
Source link