Cancer research with tremendous promise for breakthroughs
[ad_1]
A research team led by Gary Ying Wai Chan from the School of Biological Sciences at The University of Hong Kong (HKU), has unraveled the function of a unique enzyme, ANKLE1.
ANKLE1 acts on chromatin bridges trapped in the midzone of dividing cells. By cutting this bridge, ANKLE1 prevents damage to genetic material and stops the immune system from attacking the body’s own cells by mistake.
Understanding the mechanisms by which chromatin bridges are cleaved during cell division is important for developing new strategies to prevent or treat diseases such as cancer and autoinflammatory disorders. This research is highly relevant to cancer and auto inflammation and was recently published in Advanced Science.
Background
During cell division, DNA must be properly divided into two new cells. However, mistakes can occur during this process, causing DNA sections to stick together and form structures called chromatin bridges. This bridge is like a strand of DNA that connects two separate masses of chromosomes in the new cell. As a result, like it or not they will get stuck in the middle of the cell division process.
When chromatin bridges become trapped, they are broken by contractile forces mediated by the actin-myosin complex, which can lead to DNA damage and the formation of small nuclei called ‘micronuclei’, causing genomic instability, characteristic of many solid tumors, and activating an innate immune response that contribute to auto-inflammation.
To investigate alternative mechanisms by which cells complete chromatin bridges, a team of Chinese researchers recently discovered that ANKLE1, a unique endonuclease (enzyme), plays an important role in cleaving chromatin bridges in the midsection during cell division, thereby avoiding damage from mechanical damage. . strength. This alternative mechanism helps ensure proper segregation and stability of DNA, which is important for preventing diseases such as cancer and autoinflammatory disorders.
Role of ANKLE1 in preventing DNA damage and autoimmunity
Previously, scientists identified an endonuclease called LEM-3 Caenorhabditis elegans, a microscopic roundworm commonly used as a model organism in biological research. LEM-3 has been found to play a role in completing chromatin bridges in the ‘middle body’, a structure that connects two daughter cells during cell division. The midsection is eventually cut off, a process known as abscission, which results in the complete separation of the two cells.
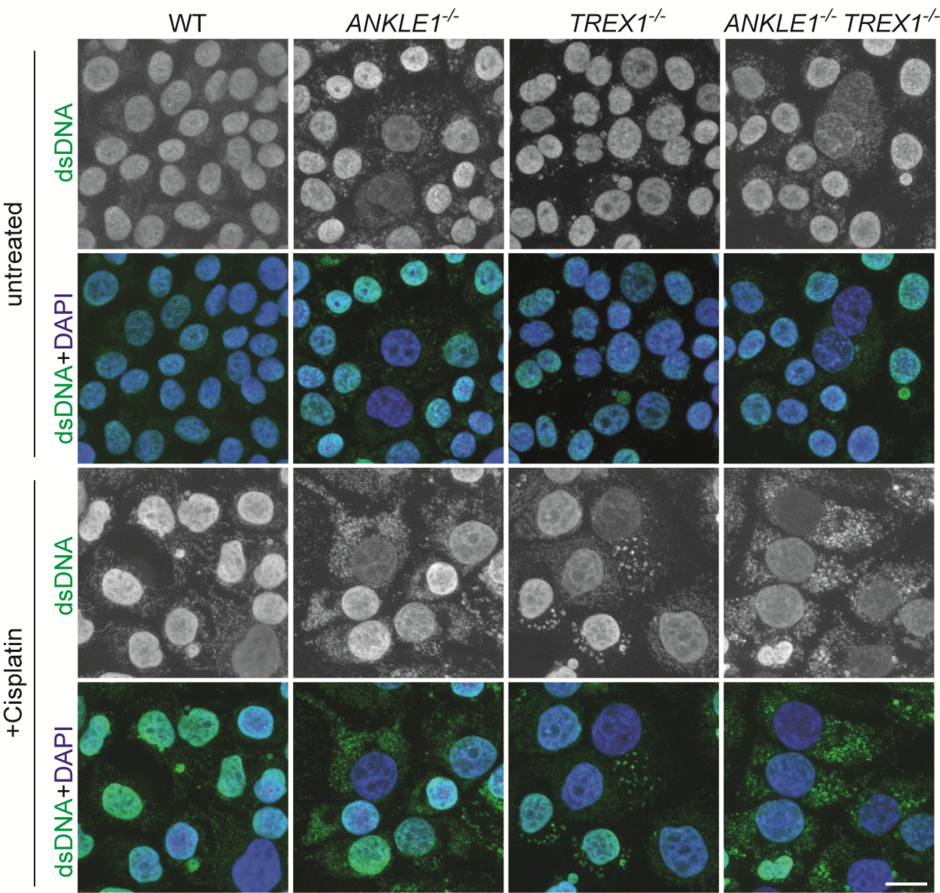
To understand the cellular function of ANKLE1, the human equivalent of LEM-3, the research team used CRISPR/Cas9 genome editing technology to delete the ANKLE1 gene in human cells.
During the process, the team found that loss of ANKLE1 leads to increased formation of extended chromatin bridges, suggesting that cleavage of the bridge by ANKLE1 prevents further DNA stretching. In the absence of ANKLE1, the persistent bridge will eventually be broken by the contractile forces of the actomyosin separating the two daughter cells. This mechanical damage induces massive DNA fragmentation, resulting in micronucleus formation and DNA release into the cytosolic DNA, which can trigger an immune response and lead to inflammation.
Importantly, loss of ANKLE1 not only induces DNA damage and genomic instability, but also leads to robust activation of cGAS-STING innate immunity. The cGAS-STING pathway is the primary innate immune defense system against pathogens, as it senses the presence of DNA in the cytosol. In the absence of ANKLE1, DNA fragments are generated at bridge breaks and the resulting cytosolic DNA is misinterpreted as pathogenic DNA. These results indicate that ANKLE1 plays a role in maintaining genome stability and preventing autoimmunity.
Research implications
The researchers say this study improves our understanding of the relationship between chromatin bridges and the innate immune response.
“More and more research data shows that the immune response plays an important role in determining the clinical outcome of many traditional anti-cancer drugs,” said Dr Gary Ying Wai Chan.
“For example, the use of antimitotic drugs is a common strategy for treating solid tumors. However, whether the induction of the immune response due to the resulting chromatin bridges is partly responsible for the antitumor effect remains unclear. The link between chromosomal instability and innate immunity is sure to be an area of cancer research with tremendous promise of breakthroughs.”
[ad_2]
Source link