Nanotechnology designs open doors for new cancer treatments
[ad_1]
(Nanowerk News) To develop cancer drugs, researchers often have to make difficult tradeoffs. Most drugs that are potent enough to kill tumors also cause unpleasant side effects – and may end up doing more harm than good. Now, researchers and clinicians at Stevens Institute of Technology and Hackensack Meridian Health have developed a new drug delivery system that uses gold nanoparticles to deliver drugs to tumors with incredible precision—while dramatically reducing potential systemic side effects.
In a paper published in ACS Applied Materials & Interfaces (“Targeted Lymphoma Therapy Using a Gold Nanoframework-Based Drug Delivery System”), the team describes the ability to package and seal drugs into a porous gold nanoframe, then apply a layer of hyaluronic acid to the outer surface of the nanoparticles to further seal the drug inside. The result: extremely stable particles that only release their charge when they bind to hyaluronic acid receptors on the surface of lymphoma tumors.
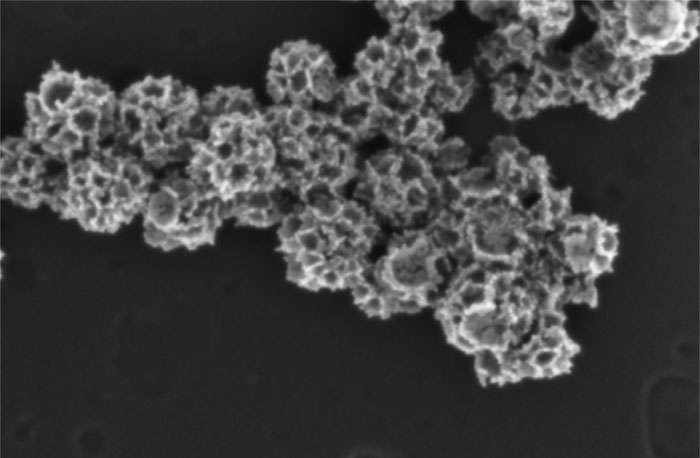
“Gold is very stable, so it’s the perfect material for drug delivery,” said Hongjun Wang, a professor of biomedical engineering and director of the Semcer Center for Healthcare Innovation at Stevens who led the research with Johannes Zarkzweski, a physician at Hackensack. . “Using this approach, we can deliver drugs more precisely and get much better clinical outcomes.”
Because drugs are administered selectively, with minimal leakage into the bloodstream, much lower amounts of drug can be used to treat tumors, reducing the risk of toxic side effects. In animal studies, the team found no sign of toxicity and no detectable levels of the drug in the bloodstream, despite the fact that the chemicals they administered typically caused detectable toxicity in 50 percent of subjects.
Gold nanoparticles continue to circulate in the bloodstream much longer than free-floating drug molecules, meaning that patients currently receiving daily treatments may be able to switch to weekly or biweekly regimens. In animal trials, lymphoma tumors also responded to targeted administration of drugs markedly better than non-targeted treatments, indicating that new treatment protocols could be developed to improve outcomes in human cancer patients.
Using gold as a delivery system opens the door for other clinical applications, including the use of tumor-seeking gold nanoparticles as a contrast agent to enable more precise imaging. The particles can also be heated by shining harmless light energy into the patient’s body, effectively “cooking” the tumor from within. They can also be calibrated to deliver their pharmaceutical payload only when triggered by incoming light, providing additional control over how and when drugs are delivered.
Nanoparticles can be filled with drugs very quickly and efficiently, using a chemical process that is cheap and scalable — a fact that should help offset the cost of using gold for shipping drugs. “Cost is always a concern, of course,” said Wang. “But it’s a simple methodology, and we think the benefits speak for themselves.”
While the current study focuses on validating the use of gold nanoparticle drug delivery systems in animals, Wang hopes to see the approach developed for testing in human cancer patients. “We’ve shown it works in animals – now we need to show it’s safe in humans too,” he explained. Pharmaceutical companies are already showing interest in the approach, and the FDA has approved the use of gold in other clinical applications, he said.
Meanwhile, Wang added, the main research goal is to develop more advanced outer pore sealants that rely on antibodies to target other, more difficult-to-treat tumors with similarly high specificity. “Our ultimate goal is to use this approach to target multiple myeloma and other cancers that are currently incurable,” said Wang. “I hope we can complete the basic research and push this into clinical trials soon.”
[ad_2]
Source link