A new way to target sepsis and metastases through common molecular targets
[ad_1]
(Nanowerk Highlights) Cells have an innate ability to adapt quickly to chemical and physical stimuli of the environment. Although genetic mutations can cause changes in cellular properties, the more rapid responses are often driven by non-genetic mechanisms. This cellular agility, otherwise known as cell plasticity, plays a critical role in the orchestration of fundamental biological processes, both in health and disease.
Consider, for example, the dynamic properties of tumor cells. These cells can transition from a highly proliferative state to a highly invasive state, thereby facilitating cancer spread, or metastasis. Similarly, during an episode of inflammation, immune cells undergo transformation, becoming active agents in the execution of the inflammatory response and the promotion of subsequent tissue repair. However, when this inflammatory response gets out of control, it can trigger tissue damage and even lead to a life-threatening condition known as septic shock.
A research team based at the Curie Institute in Paris recently unveiled a new molecular actor in this process: copper. Their findings, published in the journal Natural (“Anesthetic copper signaling pathways that drive inflammation”) revealed that the underlying processes driven by copper are consistent across inflammation and cancer metastasis.
Given the global health burden of this condition – with more than 11 million annual deaths from septic shock and 90% of cancer-related deaths caused by metastases – discovering the role of copper could be a significant breakthrough. This new understanding of the influence of copper on cellular plasticity may pave the way for innovative therapeutic strategies in the future.
Inflammation caused by copper can be stopped with supformin
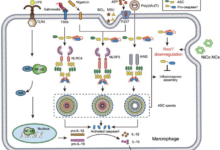
By analyzing metal homeostasis in immune cells called macrophages and cancer cells, the research team found that aggressive inflammatory cells and cancer cells displayed high levels of copper. Scientists designed a new molecule based on metformin, which can block this reaction by binding to copper.
Interestingly, these findings harken back to research from 1929 which first revealed metformin’s ability to bind copper, despite forming a 2:1 complex with two metformin molecules. The newly engineered molecule, named supformin, combines two metformin units, thereby increasing the capacity to form copper complexes. Impressively, supformin displays efficacy that is 5000 times greater than its metformin counterpart.

This new drug specifically targets mitochondria, the cellular powerhouses responsible for energy production. Here, copper serves as a catalyst in the interconversion of the vital enzymatic cosubstrate, NAD(H).
Inside the mitochondria, supformin binds to this copper to inactivate this metabolic process. NAD(H) normally triggers the Krebs cycle, the main energy supply pathway in cells. By blocking NAD(H) generation, supformin triggers the reduction of a key metabolite that is important for facilitating epigenetic changes. It is a non-genetic modification that changes the cell phenotype.
This results in a less inflammatory state in the immune cells and a less aggressive state in the cancer cells. The therapeutic potential of supformin was demonstrated in several rat models of sepsis, where administration of supformin greatly increased the survival rate of the animals.
How does copper get into the cell?
In this process that underlies cell plasticity, copper is taken up by cells via a protein called CD44 and hyaluronic acid, which are also known ingredients in many beauty products. There is already evidence of metal uptake by CD44 in cancer cells by the research team, which was previously published in the journal Natural Chemistry (“CD44 regulates epigenetic plasticity by mediating iron endocytosis”).
CD44 is a protein that has been studied extensively for decades and is found in many types of cells, including immune system cells, cancer cells, cells involved in wound healing, red blood cell progenitor cells and many more. Scientists showed that the copper taken up by CD44 accumulates in mitochondria.
What’s the bigger picture?
This work provides a mechanistic understanding of cellular state control, illustrating how modifications in energy metabolism can induce epigenetic changes that influence gene expression. Historically, metals have been somewhat underestimated in their cellular role, often being thought of simply as cofactors. However, given that many metals are powerful catalysts in the physical world, and given that life has evolved within the confines of this physical world, they must be recognized as essential cellular components.
Our results for iron and copper place these metals at the center of biological attention. These findings change our understanding of how inflammation and metastatic formation in cancer are regulated and provide new ways to intervene therapeutically. As a result, there is potential for the development of new drugs for a wide variety of conditions, including septic shock and various forms of cancer.
This work is revolutionizing the way we consider changes in gene expression, putting mitochondria into the picture as cell organelles that control the way cells behave. Supformin’s promise as a therapeutic agent now needs to be validated through rigorous testing and potential drug development processes.
Additionally, given that supformin, which is based on metformin, targets mitochondrial copper, this study also demonstrates the general mode of action of the widely used drug metformin. This may provide an explanatory framework for the diverse phenotypes and effects associated with metformin use, as previously documented in the literature.
Become a Spotlight guest writer! Join our large and growing group of guest contributors. Have you recently published a scientific paper or have other interesting developments to share with the nanotechnology community? Here’s how to publish on nanowerk.com.
[ad_2]
Source link