Algae combined with visible light can produce ink for cultured meat
[ad_1]
(Nanowerk News) A few years ago, a French daily newspaper published an article entitled “Utilization of Algae in Korea as a Foodstuff for the Earth”. The article highlights the ecological potential of algae, which Westerners usually find unattractive due to their soft, mushy consistency. Algae have the ability to absorb atmospheric carbon dioxide and produce far less carbon emissions. Consequently, the act of consuming algae alone can play a constructive role in environmental protection.
Despite this, progress has been made in the production of meat grown using algae, thereby presenting a new approach to conserving the Earth. In addition, algae can also be used in engineering artificial organs for those who experience organ failure.
The research team led by Professor Hyung Joon Cha of the Department of Chemical Engineering and the Convergence School of Science and Technology, and PhD candidates Sangmin Lee and Dr. Geunho Choi of the Department of Chemical Engineering at POSTECH has developed a bioink characterized by improved cells. survival and printing resolution. This achievement was achieved through the utilization of alginate derived from algae, natural carbohydrates and harmless visible light.
The research results have been published in Carbohydrate Polymers (“A tyramine-conjugated light-crosslinkable alginate-based microgel bioink for multiple cell-laden 3D artificial organs”), international journal in the field of biomaterials.
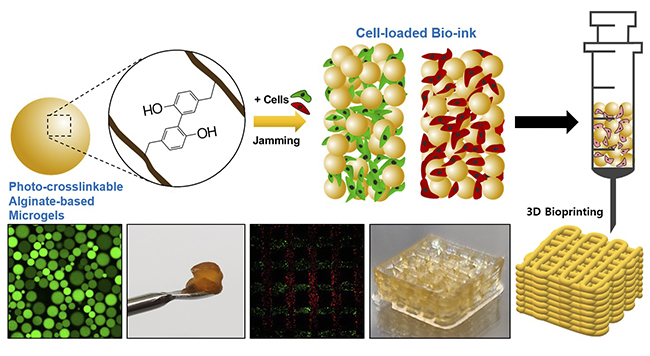
3D bioprinting is a methodology used in the manufacture of artificial organs or tissues through the use of cell-containing bioinks1). This technique holds great promise in the fields of tissue engineering and regenerative medicine while attracting a lot of attention in the food technology sector because of its potential to produce cultured meat, an emerging concept in future food production. Nevertheless, currently available bioinks exhibit limitations, inhibiting cell mobility and resulting in low cell viability and printing resolution.
To overcome this challenge, the research team created microgels using photocross-linkable alginate2). Later, they developed a 3D printed bioink capable of facilitating free cell movement and proliferation by using these photocrosslinkable alginate microgels. These cell-laden microgel bioinks led to a 4-fold increase in cell proliferation compared to conventional bioinks. In addition, microgels exhibit reduced viscosity when subjected to external forces over a period of time, quickly recovering their initial shape even after deformation. These characteristics significantly increase the resolution and lamination capacity of printed output.
Professor Hyung Joon Cha who led the research explained, “We engineered the structure of the functional network using bioink-based biomaterials with excellent and stable cell loading capabilities for practical 3D printing.” Expressing optimism for the future, he added, “Future research and refinement of this technology is expected to promote its wide application in artificial organ engineering and cultured meat production.”
[ad_2]
Source link