Quantum effects were detected in the collision of hydrogen and noble gases
[ad_1]
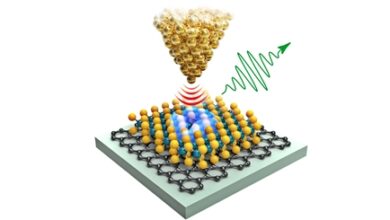
(Nanowerk News) A team of Freie Universität Berlin researchers led by quantum physicist Professor Christiane Koch have demonstrated how the hydrogen molecule behaves when it collides with an atom of a noble gas such as helium or neon.
In an article published in the journal Science (“Feshbach resonance state tomography”), the researchers explain how they used simulation to draw connections between data from experiments and theoretical models of quantum physics.
The study includes theoretical calculations as well as data collected in experiments with atoms and molecules carried out at the TU Dortmund University and the Weizmann Institute of Science in Israel. The team was able to show that the collisions change the way molecules vibrate and rotate under the laws of quantum mechanics. Research in the field of quantum mechanics continues to be important in today’s world. Findings like these have applications in the development of cell phones, television, satellite and medical diagnostic technologies.
The quantum effect observed here is known as the Feshbach resonance. “For a moment after the collision, the hydrogen molecule and the noble gas atom form a chemical bond and then separate again,” explains Professor Koch of the Freie Universität Berlin.
However, despite very detailed measurements and calculations for relatively small and simple systems, the researchers are still far from being able to reconstruct the full quantum mechanical characteristics of the noble-hydrogen gas collision. “This is due to one of the fundamental phenomena of quantum mechanics: when it comes to measurements, you cannot escape the basic principles of classical physics. That creates a dilemma: We can describe certain quantum mechanical phenomena mathematically in abstract terms, but still need to use concepts from classical physics to fully understand them,” explains Koch.
Quantum effects – meaning a type of behavior that cannot be explained by the rules of classical physics – arise when atoms and molecules can no longer be adequately described by the places they occupy and the speed at which they move.
“They display characteristics that we associate with wave dispersion, such as interference, meaning the constructive or destructive layering of waves,” said Koch.
In addition, there is another phenomenon such as entanglement, which occurs when quantum mechanical objects exert a direct influence on each other despite being spatially distant.
Quantum effects usually appear in the realm of very small objects such as atoms and molecules, and when these objects are under the slightest influence from their environment. The latter is achieved for very short bursts of time or under very low temperatures close to absolute zero (-273.15 °C).
“In this state, only a small number of so-called quantum states are available to these particles. Systems basically behave in an orderly way,” said Koch.
Higher temperatures allow for a greater number of quantum states in a particle, and quantum mechanical effects tend to flatten out when distributed as a statistical average across states, and thus essentially disappear from view. In this state, the system behaves more randomly and can be described using statistics. So far, even the coldest atomic-molecule collisions have exhibited this statistically predictable behavior.
“That makes it almost impossible to arrive at any conclusions about the interactions between atoms and molecules, which means we can’t make direct connections between real-life experimental data and theoretical models,” explained Koch.
[ad_2]
Source link