Sahmyook University Researchers Examine Ultra-High Stability Nano Bubbles
[ad_1]
Nanobubbles (NBs) are extremely valuable for industrial, agricultural and water treatment applications because of their exceptional stability. However, the mechanisms underlying such stability are unclear. To address this knowledge gap, researchers from South Korea investigated the stability of high concentrations of sub-200-nm NBs under various conditions, finding that they exhibited remarkable stability even after exposure to temperature variations and severe physical impact. NB, therefore, could find potential applications in mass production and distribution in bubble technology.
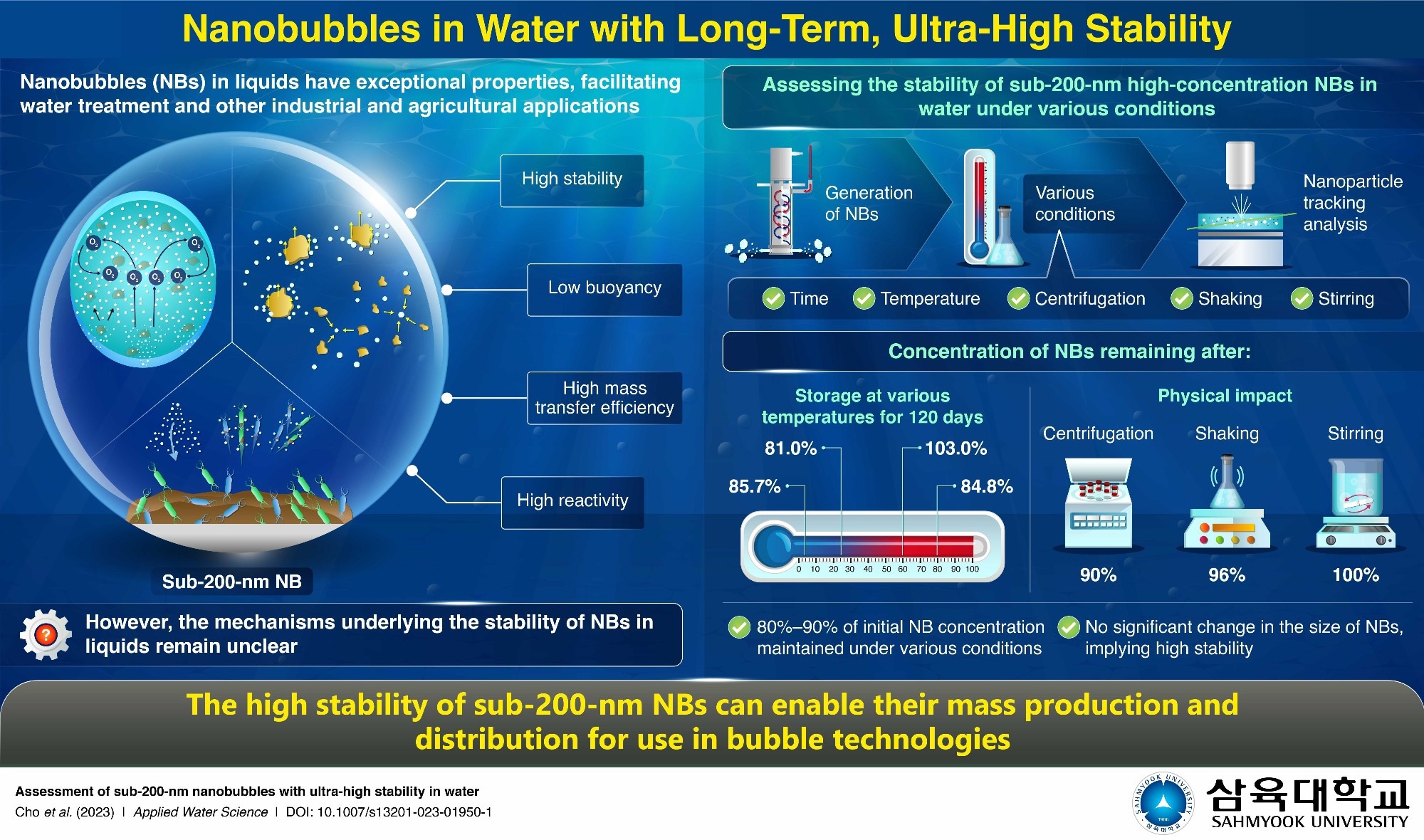
Image Credit: Sahmyook University
Bubble technology has emerged as a powerful tool for tackling environmental pollution, improving water treatment processes, and increasing industrial and agricultural production. New applications of this technology have emerged due to the unique properties of nanobubbles (NB)—gas bubbles smaller than 1,000 nanometers (nm) in diameter. In particular, NBs in water, especially those less than 200 nm in diameter, exhibit low buoyancy, high mass transfer efficiency, high reactivity and outstanding stability. However, the underlying mechanisms behind their stability remain elusive, with most studies focusing solely on temporal changes in the size and surface charge of NBs and ignoring changes in their concentrations under various conditions.
To address this issue, a research team led by Associate Professor Myoung-Hwan Park from Sahmyook University in South Korea recently investigated the amount and stability of high concentrations of NB in water under various conditions. Their study was available online on 09 June 2023 and published in Volume 13, Issue 7 of the journal Applied Water Science.
Highlighting the importance of NB, Dr. Park commented: “The most promising consequence of using NBs is that they can enhance the native performance of various components without chemical additives.”
The researchers first produced air-in-water NB using custom-built NB generators, with more than two billion NB per ml of water, each measuring about 100 nm. They analyzed the stability of the NB using nanoparticle tracking analysis, which involves shining a laser on nanoscale particles suspended in a liquid and tracking their movement under a microscope. This technique allows researchers to investigate how the amount and size of NB changes under different conditions, including storage at various temperatures and exposure to physical impacts such as centrifugation, shaking and stirring.
They found that NB retained 80-90% of their initial concentration under all conditions tested. Specifically, when stored at 5 °C, 25 °C, 60 °C, and 80 °C for 120 days, NB retained 85.7%, 81.0%, 103%, and 84.8% of its initial concentration, respectively. Moreover, when subjected to centrifugation for 90 minutes, NB retained more than 90% of its initial concentration, and after eight hours of shaking, the corresponding value was 96%. Stirring the NB solution for eight hours also did not change its concentration. In addition, NB did not show any significant size change in any of the tests above.
These findings demonstrate that sub-200-nm NB exhibits exceptional stability under a wide range of conditions. “NB demonstrates significant potential for real-life applications in the mass production and distribution of bubble technology in various fields, such as pharmaceuticals, cosmetics, hygiene, environment, food, agriculture and others,” highlights Dr. Park. “In addition, scientists are working to reduce reliance on hazardous but indispensable chemicals, and the use of non-hazardous gases and NB can further support their efforts,” he concluded.
This study can thus pave new avenues for bubble technologies that offer promising prospects for safer environments and increased efficiency in water treatment, industry, agriculture and beyond.
Source: https://www.syu.ac.kr/eng/
[ad_2]
Source link