Triastek positive results of printed treatment for ulcerative colitis
[ad_1]
Triastek, Inc. has completed its first human study of its third 3D printing medicinal product, T21, designed to treat moderate to severe ulcerative colitis (UC).
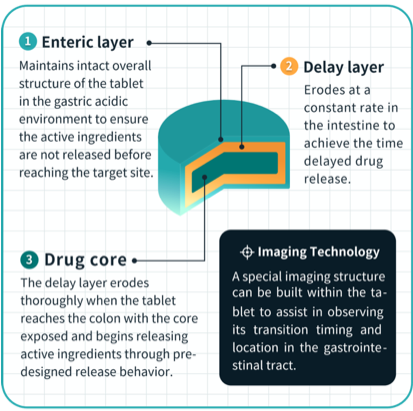
Imaging results from the study confirm that the T21 tablets are precisely delivered and released to the target site – the large intestine – for drug release.
Oral drug administration is generally considered the method of choice for patients with UC because of its safety, pain avoidance and patient compliance. However, historically it has been difficult to achieve proper drug delivery to the large intestine, without releasing any drug in different parts of the digestive tract.
Triastek developed the T21 in an effort to solve the problem of precise, targeted delivery of oral drugs to the gastrointestinal tract. Through the company’s 3D Melt Extrusion Deposition (MED) printing process, its 3D printed tablets control when, where, and how much drug is released in the digestive tract, ensuring more targeted and efficacious drug delivery.
“The first in human study data with T21 verified the precise colon delivery capability of the MED process, and the platform is poised to become the new drug delivery system of choice for new products targeting the colon with local efficacy or systemic absorption,” said Xiaoling Li, co-founder and chief scientific officer of Triastek.
“We look forward to continuing to demonstrate how Triastek’s 3D printing process can provide pharmaceutical companies with technical solutions for efficient product development of optimized drug delivery, ultimately leading to the ability to provide drugs of more clinical value to patients.”
Triastek aims to develop partnerships with pharmaceutical companies to help them differentiate their products through the use of 3D drug printing. Triastek recently partnered with Boehringer Ingelheim, Eli Lilly and Merck KGaA.
About T21
T21 is Triastek’s third 3D printed drug product, and is a colon-targeted oral delivery drug for moderate to severe ulcerative colitis (UC). In November 2022, Triastek received approval for Investigational New Drug (IND) application for T21 from the United States Food and Drug Administration (FDA).
The original drug was an oral Janus kinase (JAK) inhibitor called tofacitinib. Following the approval of T21, Triastek is launching the FIH study in Q1 of 2023 to verify the transport, attrition process and site of release of T21 in the human body after oral administration. To date, T21 has completed its first study in humans, and imaging results have confirmed that T21 tablets are delivered accurately to the target site – the large intestine.
About Melt Extrusion Deposition (MED)
Melt Extrusion Deposition (MED) 3D printing is an additive manufacturing, end-to-end process that continuously converts powdered raw materials into a soft/liquid state followed by precise layer-by-layer deposition to produce objects with well-designed geometric structures.
This optimized product design helps control when, where, and how much drug is released in the digestive tract, ensuring more targeted and efficacious drug delivery. The MED process can be used to design and develop very different new chemical entities and life cycle management products for unmet medical and clinical needs.
[ad_2]
Source link