Scientists develop artificial molecules that behave like real ones
[ad_1]
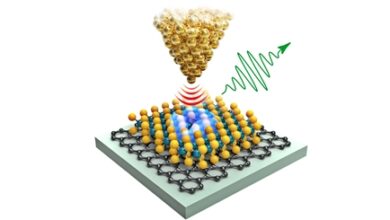
(Nanowerk News) Scientists from Radboud University have developed synthetic molecules that closely resemble real organic molecules. The research collaboration, led by Alex Khajetoorians and Daniel Wegner, can now simulate the behavior of real molecules using artificial molecules. In this way, they can change the properties of molecules in ways that would normally be difficult or unrealistic, and they can better understand how molecules change.
Emil Sierda, who is in charge of conducting experiments at Radboud University: ‘A few years ago we had the crazy idea to make a quantum simulator. We want to make artificial molecules that look like real molecules. So we developed a system where we can trap electrons. The electrons surround the molecule like a cloud, and we use the trapped electrons to build artificial molecules.’ The results the team found were astounding. Sierda: ‘The similarities between what we built and real molecules are uncanny.’
The research has been published in Science (“Quantum simulator for emulating low-dimensional molecular structures”).
Change molecule
Alex Khajetoorians, head of the Scanning Probe Microscopy (SPM) department at Radboud University’s Institute of Molecules and Materials: ‘Making molecules is quite difficult. What is often more difficult, however, is understanding how certain molecules react, for example how they change when they are twisted or twisted.’ How molecules change and react is the basis of chemistry, and leads to chemical reactions, such as the formation of water from hydrogen and oxygen. “We want to simulate molecules, so we can have the ultimate device to bend them and tune them in ways that are nearly impossible with real molecules.” That way we can say something about real molecules, without building them, or without having to deal with the challenges they present, such as their changing shape.’
Benzene
Using this simulator, the researchers created an artificial version of one of the basic organic molecules in chemistry: benzene. Benzene is a starting component for a large number of chemicals, such as styrene, which are used to make polystyrene.
Khajetoorians: ‘By making benzene, we simulated a textbook organic molecule, and built a molecule composed of elements that are not organic.’ On top of that: the molecule is 10 times larger than its native counterpart, which makes it easier to work with.
Practical use
The uses for this new technique are endless. Daniel Wegner, assistant professor in the SPM department: ‘We’re just starting to imagine what we could use this for. We have so many ideas it’s hard to decide where to start.’ Using simulators, scientists can better understand molecules and their reactions, which will help in every scientific field imaginable.
Wegner: ‘New materials for future computer hardware are very difficult to manufacture, for example. By creating simulated versions, we can look for new properties and functions of certain molecules and evaluate whether it is feasible to make real materials.’
In the far future, all sorts of things may be possible: understanding chemical reactions step by step as in slow-motion video, or creating artificial single-molecule electronic devices, such as shrinking the size of a transistor on a computer chip. Quantum simulators are even suggested to work as a quantum computer.
Sierda: ‘But that’s a long way off, for now we can start by starting to understand molecules in ways we haven’t understood before.’
[ad_2]
Source link