A safe, easy and affordable way to store and retrieve hydrogen
[ad_1]
(Nanowerk News) Researchers at the RIKEN Center for Emergent Matter Science (CEMS) in Japan have discovered a compound that uses a chemical reaction to store ammonia, potentially offering a safer and easier way to store this important chemical.
These findings, published in Journal of the American Chemical Society (“Ammonia storage chemistry via dynamic structural transformation of a hybrid perovskite compound”), allows not only to store ammonia safely and conveniently, but also to transport important hydrogen. These findings will help lead the way to a decarbonized society with a practical hydrogen economy.
For society to move from carbon-based energy to hydrogen-based energy, we need safe ways to store and transport hydrogen, which is itself highly flammable. One way to do this is to store it as part of other molecules and extract it as needed. Ammonia, chemically written as NH3, makes a good hydrogen carrier because three hydrogen atoms are packed into each molecule, with nearly 20% of the ammonia being hydrogen by weight.
The problem, however, is that ammonia is a very corrosive gas, making it difficult to store and use. Today, ammonia is generally stored by thawing it at below freezing temperatures in pressure-resistant containers. Porous compounds can also store ammonia at room temperature and pressure, but their storage capacity is low, and the ammonia cannot always be recovered easily. The new study reports the discovery of perovskite, a material with a characteristic repeating crystal structure, which can readily store ammonia and also allows easy and complete uptake at relatively low temperatures.
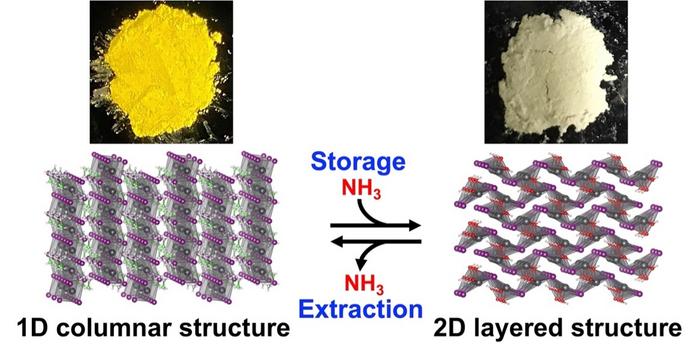
The research team led by Masuki Kawamoto at RIKEN CEMS focused on perovskite ethylammonium lead iodide (EAPbI3), is chemically written as CH3CH2NH3PbI3. They found that its one-dimensional columnar structure undergoes a chemical reaction with ammonia at room temperature and pressure, and dynamically transforms into a two-dimensional layered structure called lead iodide hydroxide, or Pb(OH)I.
As a result of this process, ammonia is stored in a layered structure through chemical conversion. So, EAPbI3 can safely store corrosive ammonia gas as nitrogen compound in a process that is much cheaper than liquefaction at -33 °C (-27.4 F) in a pressurized vessel. More importantly, the process for recovering stored ammonia is just as simple.
“To our surprise, the ammonia stored in ethylammonium lead iodide can be easily extracted by gently heating it,” said Kawamoto. Stored nitrogen compounds undergo a reverse reaction at 50 °C (122 F) under vacuum and return to ammonia. This temperature is much lower than the 150 °C (302 F) or more required to extract ammonia from porous compounds, making EAPbI3 an excellent medium for handling corrosive gases in a simple and cost-effective process. Additionally, upon returning to a one-dimensional columnar structure, the perovskite is reusable, allowing ammonia to be stored and extracted repeatedly. An added bonus is that the normally yellow compound turns white after the reaction. According to Kawamoto, “the ability of compounds to change color when storing ammonia means that color-based ammonia sensors can be developed to determine the amount of ammonia stored.”
The new storage method has several uses. In the short term, researchers have developed a safe method for storing ammonia, which has many uses in society, from fertilizers to pharmaceuticals to textiles. “In the long term,” said co-author Yoshihiro Ito of RIKEN CEMS, “we hope that this simple and efficient method can be part of the solution to achieve a carbon-free society through the use of ammonia as carbon-free hydrogen. carrier.”
[ad_2]
Source link